Top EMS Products
-
-
-
-
-
Respiratory
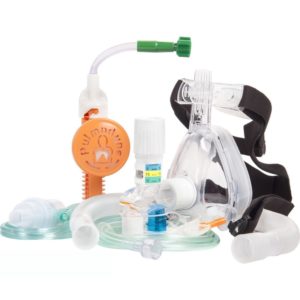
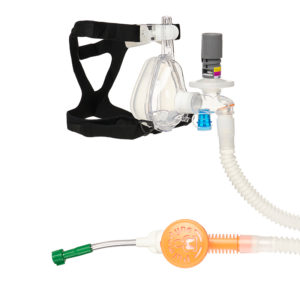
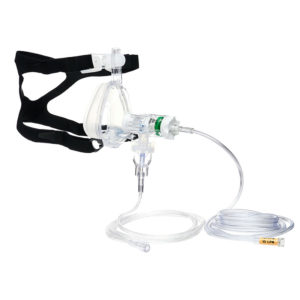
CPAP, Pulmodyne, O2-MAX BiTrac ED Mask, 5-SET Valve, Expandable Tubing, Filter,
From: $82.95 -
-
-
Penn Care - Your Emergency Medical Solution Provider
Welcome to Penn Care. We are not a large conglomerate; in fact, we have fewer than 50 employees. We are, however, a small but mighty team with a firm belief in core values backed up by hard work from a well-trained and dedicated staff that relentlessly strives for perfection.
Our customers are the reason for our success, and each year we work hard to improve what we do so that they benefit from our constant evolution. Change is in our culture and we take pride in that.
Are you searching for a provider that will do what it takes to solve your problems? Penn Care is the place! In need of a partner company that knows who you are? Give us a call!
Penn Care is ready to assist with all of your emergency medical needs: EMS Supplies, Ambulance Sales, EKG Monitors, and Disaster Response Equipment.ukmeds.co.uk